
Metal Colouring and Patination with
Solutions from e-waste
For six months I acted as an artist in residence for one day a week (2017 - 2018) at the Love Chemistry Group part of Edinburgh University. I employed their hydrometallurgy metal recovery process with the aim of reclaiming some copper and gold from electronic waste. Hydrometallurgy is a branch of extractive metallurgy that uses aqueous chemistry to recover metals from ores, concentrates, and recycled materials. At this time the group employed acid-based solutions for metal recovery but have since moved on to using alcohol solutions. Working with metals in solution suggested to me the potential to use these for metal colouring and patination. This chemical approach to metalworking is in contrast to the traditional mechanical approach that most jewellers and silversmiths will be familiar with.

I purchased some computer circuit board fingers from e-bay that had gold rich tips. I started by trimming the gold rich elements leaving the rest of the board. Working with the largest 5 litre flask that the laboratory had I added my circuit board fingers to a solution of hydrochloric acid. This was to remove all the metals from the fingers. The mixture was agitated for around two weeks and created a deep green solution with foam on top. The green colour was not a great surprise as copper is the predominant metal in e-waste. One of the PhD students I was working with called this process ‘molecular gold panning’! The image above shows the circuit board fingers before and after the metal recovery process. This process also reminded me of the old Blackadder sketch where the character Percy is trying to be an alchemist and create some gold but instead has created ‘purest green’.



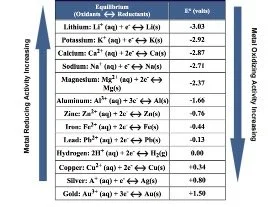
My initial experiments involved working with a chemical reduction and oxidation (redox) process where metal in solution was applied to pieces of sterling silver sheet to see how it would react. This is called ‘electrochemistry’. This is a branch of chemistry that looks at ‘reduction’ and ‘oxidation’ reactions, commonly referred to as ‘redox reactions’ where the electrons of one metal compound in solution will be lost while the other solid metal will gain electrons. This exchange of electrons creates a force measured in volts hence the name electrochemistry. We know from the electrochemical series, that is the list of metals arranged in order of how easily the metal atoms lose electrons, that some metals are more reactive than others. For example, silver ions are oxidising, therefore should oxidise copper (remove electrons) to form copper ions and, at the same time, deposit silver metal. But this wouldn’t work the other way around – ie copper will not deposit onto silver as it is more reactive than silver.
I experimented with droplets of different solutions from the metal recovery process on small pieces of different silver alloys (fine 999, sterling 925, and Britainnia 956) to see what effects could be achieved. The first solution containing all of the metals recovered (Copper, gold, cobalt, iron etc) created a beautiful effect, with the copper (the largest quantity of metal extracted) contained in the solution crystallizing around the edges as it dried out. These droplets were around one centimeter in diameter. I created a time-lapse video of this process, in which around three-and-a-half hours is condensed into a one-minute sequence. This result is fairly robust and does not break off the silver sheet easily. This is a way of colouring metals using one metal solution to plate another metal, for example sterling silver sheet, without the need for electricity. This technique also creates an even plating of one metal on the other.



Close up images of the droplets of e-waste solutions on pieces of sterling silver sheet. Experiments with a weak gold chloride solution on different types of silver and found that on sterling silver the gold crystallized on the surface of the silver but could easily be brushed off. On fine silver, it was more robust, creating a lovely milky effect that did not brush or rub off. A strong gold chloride solution created a beautiful deep brown surface finish, with veins of gold visible on all of the silver alloys.
I subsequently discovered from my involvement in another research project for the British Museum documenting silversmithing practices in Ecuador that this redox technique is also known as electrochemical replacement plating and was commonly practiced in ancient times in the Andes. Sometimes also referred to as electrochemical displacement plating this was a sophisticated, pre-Columbian Andean technique, primarily developed by Moche craftsmen (c. 200–900 CE) to gild or silver copper objects. It involved dipping cleaned copper into an acidic, corrosive mineral solution containing gold or silver, allowing the precious metal to plate onto the surface without electricity. This is something anthropologist and materials scientist Heather Lechtman has written about https://www.jstor.org/stable/3104667


Hand raised sterling silver dish with sim card imprint and stripe of e-waste mixed metals solution applied using electrochemical displacement plating (redox process).
Sandra Wilson 2019, Sterling Silver. Image: Diarmid Weir
Very little gold was recovered from this first metal recovery process so I created a series of exemplar pieces using a gold chloride solution. These exemplars replicate the same outcomes had sufficient gold solution been recovered. They demonstrate the potential for gold in solution to create novel metal colouring.

Concentrated gold chloride solution on hand raised sterling silver bowl.

Weak gold chloride solution on sterling silver pendant and pendant with embedded pieces of e-waste left over from the metal recovery process - these pieces still show elements of the copper that has not been fully recovered. The pieces of e-waste looked like little gem stones to me and so I decided to set them as if they were.
Aspects of this section have been included in conference papers for example -
EU Craft Hub - TRANSFORMATION: Effect and affect of craft in society. October 2023.
Adventure and Legacy – The Science and Technology of Textiles, Jewellery and Materials - Online, United Kingdom 11 September 2020
To read more about adapting a Japanese patination recipe using solutions from electronic waste - click this button on Kin-Furubi Patination